Breaking the Biofilm: The Holistic Sinus Rinse Protocol for Recurring Sinus Infections
There is a pattern I see constantly in my North Scottsdale rhinology practice. A patient arrives with their fourth or fifth sinus infection of the year. They have been to urgent care twice. Their primary care physician has prescribed three courses of oral antibiotics. They feel better for two or three weeks after each course — and then the infection is back. Nobody has explained to them why this keeps happening. Nobody has tested what is actually living in their sinuses. And nobody has discussed with them whether the treatment they keep receiving is even capable of reaching the bacteria that are driving their symptoms.
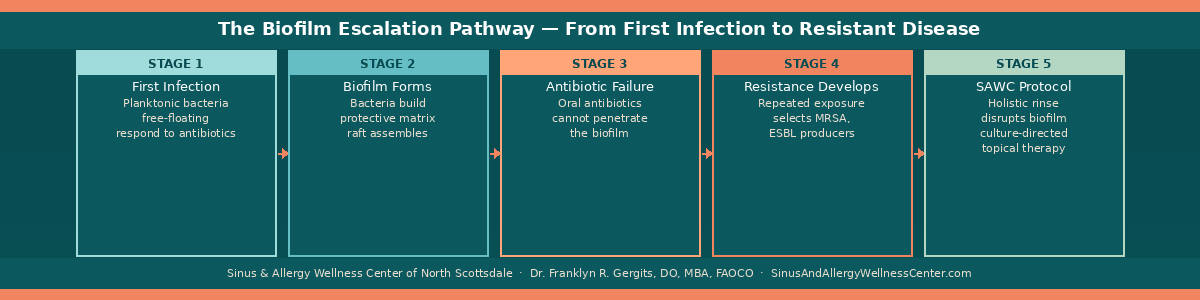
The reason antibiotics fail these patients is often not the antibiotic itself. It is what the bacteria have built around themselves — a protective structure called a biofilm — that makes the antibiotic irrelevant before it even arrives at the site of infection.
This article explains what biofilms are, why they are so common in patients with recurrent and chronic sinusitis, what we are actually finding when we culture intraoperative specimens from these patients, and what you can do about it before you ever need surgery — using three ingredients you can find today.
Clinical rhinology education written in plain language. Subscribe to the Airway & Sinus Wellness Review.
What the 2025 AAO-HNS Guideline Says — And What It Doesn’t
The American Academy of Otolaryngology — Head and Neck Surgery Foundation published its updated Clinical Practice Guideline on Adult Sinusitis in August 2025 — the first major update since 2015, incorporating evidence from 194 systematic reviews and 133 randomized controlled trials.1
The guideline defines recurrent acute rhinosinusitis (RARS) as four or more episodes of acute bacterial rhinosinusitis per year without persistent symptoms in between episodes.1 It defines chronic rhinosinusitis (CRS) as sinonasal inflammation lasting more than twelve weeks, confirmed by objective documentation — nasal endoscopy or CT imaging.1
Critically, the 2025 guideline makes a strong recommendation that clinicians should not routinely prescribe antibiotics for CRS without an acute exacerbation, and specifically states that antibiotics should not be required as a prerequisite to imaging or surgery.1 The guideline also recommends saline nasal irrigation and topical intranasal corticosteroids as the primary symptom-management tools for CRS.1
What the guideline does not address — and what remains a significant gap in standard sinusitis management — is the role of bacterial biofilms in perpetuating recurrent infection, and what to do about them outside of a surgical setting. That is the gap this protocol is designed to fill.
What Is a Biofilm — and Why Does It Matter for Your Sinuses
Think of bacteria in a biofilm like people floating on a makeshift raft in the middle of the ocean. The ocean is your sinus cavity. The sharks circling below the surface are your antibiotics — ready to kill anything they can reach. But the bacteria on the raft are protected. They are not swimming in the open water where the sharks can get to them. They have built a structure that keeps them safe.
That raft is the biofilm matrix — a complex three-dimensional scaffold made from sugars, proteins, and DNA that the bacteria secrete around themselves. Inside this matrix, bacteria communicate with each other through a chemical signaling system called quorum sensing. They coordinate gene expression, share nutrients, and collectively regulate how much of the biofilm to build and when. The matrix physically blocks antibiotics from penetrating to the bacteria living within it. And the slow-growing, metabolically altered bacteria inside a biofilm are far less susceptible to antibiotics even when they are reached — because most antibiotics target rapidly dividing cells, and biofilm bacteria have largely stopped dividing.
The result is a colony of bacteria that is up to 1,000 times more resistant to antibiotics than the same organism swimming freely in solution.2 A standard oral antibiotic course — amoxicillin-clavulanate, doxycycline, a fluoroquinolone — reaches therapeutic concentrations in the bloodstream but fails to penetrate the biofilm at the levels needed to kill the organisms within it. The patient feels better during the antibiotic course because the free-floating bacteria in the sinus secretions are reduced. But the biofilm remains. When the antibiotic is stopped, the bacteria emerge from the biofilm, repopulate the sinus, and the infection begins again.
This is not treatment failure in the conventional sense. It is a mismatch between the treatment strategy and the biology of the disease.
How Common Are Biofilms in CRS and RARS Patients
The published data is striking. A landmark study by Prince and colleagues examining endoscopically guided sinonasal cultures from 157 consecutive CRS patients found biofilm-forming bacteria in 28.6% of specimens overall — rising to 30.7% in patients with prior sinus surgery, with a statistically significant correlation between biofilm formation and the number of prior surgical procedures.3 Staphylococcus aureus and Pseudomonas aeruginosa were the dominant biofilm-forming organisms, accounting for 71% of biofilm-positive cultures.3
A separate study examining CRS without nasal polyps using scanning electron microscopy found biofilms present in 75% of patients before treatment — with only partial regression to 43% after an eight-week course of oral clarithromycin and topical steroids.4 In patients with nasal polyps, the prevalence is even higher: biofilms were identified in 97% of patients with CRS with nasal polyps, 82% of patients with CRS without nasal polyps, and 56% of controls, with biofilms most prevalent at the ethmoid bulla — the anatomical location most commonly involved in CRS.5
In patients who have been through multiple antibiotic courses — the RARS population that makes up a significant portion of my practice — the prevalence of biofilm-forming organisms is likely higher than any published study captures, because standard cultures dramatically undercount biofilm-associated organisms. Biofilm bacteria are slow-growing and difficult to culture by conventional methods. Next-generation DNA sequencing — the technology used by MicroGenDX — identifies these organisms by their genetic fingerprint rather than their ability to grow on a culture plate, which is why culture-directed therapy guided by NGS sequencing often reveals organisms that standard cultures miss entirely.
“Biofilm bacteria are slow-growing and difficult to culture by conventional methods. Next-generation DNA sequencing — the technology used by MicroGenDX — identifies these organisms by their genetic fingerprint rather than their ability to grow on a culture plate, which is why culture-directed therapy guided by NGS sequencing often reveals organisms that standard cultures miss entirely.”
What We Are Actually Culturing
The following organisms represent a sampling of what I identified in intraoperative cultures obtained last week from patients undergoing balloon sinuplasty after multiple courses of oral antibiotics. These are real patients, real cultures, and real organisms — the kind of microbial complexity that a single oral antibiotic prescription from an urgent care visit cannot address.
Cutibacterium acnes
Formerly known as Propionibacterium acnes, this gram-positive anaerobic organism is a normal skin and nasal commensal that becomes pathogenic under conditions of mucosal disruption and low oxygen — exactly the conditions created by chronic sinus obstruction. C. acnes is a confirmed biofilm former, and its biofilm architecture contributes significantly to its antibiotic resistance.6 Published systematic review data show erythromycin resistance rising from 10% in 2008 to 44% by 2024, with clindamycin resistance currently around 31%.7 Finding C. acnes in a sinus culture from a patient who has received multiple antibiotic courses is a signal that the organism has been under selective pressure and is likely not susceptible to the agents previously prescribed. Standard urgent care antibiotics — amoxicillin, azithromycin — will not reliably clear it.
Citrobacter freundii Complex
A gram-negative rod that harbors inducible chromosomal AmpC beta-lactamases — enzymes that can be expressed at low levels initially but dramatically upregulate in response to antibiotic exposure, creating resistance during treatment even when the organism initially appears susceptible.8 The IDSA 2024 guidance on antimicrobial-resistant gram-negative infections specifically flags Citrobacter freundii as an organism where resistance can emerge in approximately 20% of infections exposed to third-generation cephalosporins — even when initial susceptibility testing suggests the drug should work.9 Finding Citrobacter in a sinus culture means the standard oral antibiotics prescribed at urgent care — cephalexin, amoxicillin-clavulanate — are unlikely to be effective, and empiric treatment without culture guidance risks selecting for higher-level resistance. This is a gram-negative organism in a location — the sinuses — that most urgent care antibiotic protocols are not designed to cover.
Staphylococcus aureus
The most well-characterized biofilm-forming organism in chronic sinusitis. S. aureus is documented in over 60% of CRS cases in some series and carries the ability to produce methicillin resistance (MRSA) — which renders the entire beta-lactam antibiotic class ineffective regardless of dose or duration.10 Published CRS bacteriology data show MRSA accounting for over 60% of S. aureus isolates in revision CRS patients.10 S. aureus biofilm production is among the strongest documented in CRS — Prince et al. specifically identified S. aureus as one of the two dominant biofilm-forming organisms in their 157-patient cohort.3 An S. aureus culture from a patient with four prior antibiotic courses must be treated as potentially MRSA until sensitivity data proves otherwise. Empiric treatment with amoxicillin or a cephalosporin is not appropriate.
Enterobacter/Klebsiella aerogenes
Klebsiella aerogenes — formerly classified as Enterobacter aerogenes — is a member of the ESKAPE pathogen group, a collection of organisms specifically identified for their capacity to escape antibiotic activity through multiple resistance mechanisms.11 Like Citrobacter freundii, it carries inducible AmpC beta-lactamases and can develop resistance during treatment. It is also capable of acquiring extended-spectrum beta-lactamase (ESBL) production — a mechanism that confers resistance to virtually all penicillins and cephalosporins simultaneously.12 Finding this organism in a sinus culture from a patient with recurrent infections is a significant clinical signal. This is not a routine sinusitis organism. Its presence suggests substantial prior antibiotic pressure has altered the sinonasal microbiome in ways that standard empiric treatment cannot address.
Staphylococcus lugdunensis
A coagulase-negative Staphylococcus that is frequently underestimated clinically because it resembles the less pathogenic S. epidermidis on initial identification. However, S. lugdunensis is uniquely virulent among coagulase-negative staphylococci — it produces aggressive biofilms, carries virulence factors more characteristic of S. aureus, and has been associated with serious invasive infections. In the context of chronic sinusitis, S. lugdunensis should not be dismissed as a contaminant. It is a genuine pathogen in this setting and requires targeted therapy based on confirmed sensitivity data rather than empiric treatment.
Haemophilus influenzae
A classical acute sinusitis pathogen and one of the most common organisms in community-acquired bacterial rhinosinusitis. The critical issue with H. influenzae in the context of recurrent sinusitis is beta-lactamase production — an enzyme that inactivates amoxicillin and renders it ineffective. Approximately 30-40% of H. influenzae clinical isolates produce beta-lactamase.10 When a patient has received multiple courses of amoxicillin for what appears to be a straightforward sinus infection, and H. influenzae is ultimately cultured, there is a high probability that the organism was beta-lactamase positive throughout and was never being effectively treated. The appropriate agent — amoxicillin-clavulanate or a respiratory fluoroquinolone — was never prescribed because no culture was ever obtained.
This is not an unusual or extreme bacterial profile for patients with RARS and CRS who have been managed empirically at urgent care and primary care for months or years. This is what lives in chronically infected sinuses that have been repeatedly exposed to antibiotics — a complex, resistant, often biofilm-forming polymicrobial community that requires culture-directed targeted therapy to address, not another course of amoxicillin.
Why the Holistic Rinse Protocol Changes the Equation
If oral antibiotics cannot reliably penetrate the biofilm, the solution is to disrupt the biofilm before or instead of using antibiotics — and then to deliver any antimicrobial therapy directly to the mucosal surface rather than through the bloodstream.
The three-ingredient holistic rinse protocol I recommend for patients with RARS or early CRS who are caught in the antibiotic cycle works through three simultaneous and synergistic mechanisms. The goal is not to simply wash the bacteria away — though that is part of it. The goal is to destroy the raft before the sharks even arrive.
Ingredient 1: Xylitol
Xylitol is a natural sugar alcohol that disrupts the biofilm matrix at the structural level. It does this through two primary mechanisms. First, xylitol inhibits the adhesion of bacteria to the nasal mucosal epithelium — making it harder for bacteria to establish the initial attachment that biofilm formation requires. Second, it interferes with quorum sensing — the chemical communication system that bacteria use to coordinate biofilm construction. A bacterium that cannot communicate with its neighbors cannot build a coherent biofilm structure. The raft begins to fall apart before it can be fully assembled. Published data also demonstrate that xylitol inhibits the growth of Streptococcus pneumoniae and other common sinonasal pathogens in vitro, adding a mild direct antimicrobial effect to its biofilm-disrupting properties.
Ingredient 2: Johnson’s Baby Shampoo at 1% Concentration
This is the ingredient that surprises patients most. Johnson’s Baby Shampoo — specifically the original formulation — contains a surfactant that acts as a detergent on the lipid components of the biofilm matrix. The biofilm is not purely structural — it contains lipid membranes and hydrophobic domains that hold the matrix together. The surfactant in JBS at 1% concentration dissolves these components, mechanically disrupting the biofilm’s cohesion in a way that xylitol alone cannot achieve. The result is that the xylitol opens the structure and the JBS surfactant dissolves the membrane — together breaking apart the makeshift raft and exposing the bacteria within it to the surrounding environment. Published evidence for JBS in post-surgical CRS rinse protocols supports its safety on sinonasal mucosa at this concentration.
Ingredient 3: Manuka Honey — UMF 16+ or MGO 400+ from New Zealand Only
Once xylitol and JBS have disrupted and dissolved the biofilm matrix, the bacteria within it are now exposed — floating in the ocean without their raft. This is the moment Manuka honey acts. Manuka honey from New Zealand carries unique antibacterial properties not present in standard honey, derived from the methylglyoxal (MGO) content produced from the nectar of the Leptospermum scoparium plant. Its antibacterial mechanism operates through osmotic pressure — drawing moisture out of bacterial cells — and through the generation of hydrogen peroxide, which has direct bactericidal activity. Published in vitro data demonstrate activity against S. aureus — including some MRSA strains — as well as Pseudomonas aeruginosa and other common CRS pathogens. The rating matters: UMF 16+ or MGO 400+ is the minimum concentration that has demonstrated clinically meaningful antibacterial activity. Lower-rated honey products — or non-Manuka honey — do not carry the same active compound concentration and should not be substituted.
Even if the Manuka honey does not achieve bacterial killing — which is more likely against the more resistant organisms in the biofilm-disrupted community — the physical rinse itself washes those now-exposed bacteria out of the nasal cavity and down the drain. The raft is destroyed. The bacteria are in the open water. And whether the sharks kill them or the current carries them away, the colony that was making your sinuses their home is gone.
“The raft is destroyed. The bacteria are in the open water. And whether the sharks kill them or the current carries them away, the colony that was making your sinuses their home is gone.”
The SAWC Holistic Biofilm-Disruption Rinse — Complete Recipe
What you need:
-
NeilMed Sinus Rinse bottle — 240ml standard size
-
Distilled water — not tap water, not filtered water — distilled only
-
One NeilMed saline packet — included with the kit
-
Xylitol — food grade, pure — 1 teaspoon (approximately 4 grams)
-
Johnson’s Baby Shampoo — original formula, clear — 1/4 teaspoon (approximately 1ml — yields approximately 1% final concentration)
-
Manuka honey — UMF 16+ or MGO 400+ rating — from New Zealand — 1 teaspoon
How to prepare:
-
Warm 30ml of distilled water to body temperature — approximately 98°F — in a small cup
-
Dissolve the Manuka honey completely in the warm water first — Manuka honey is thick and will not mix evenly in cold water
-
Fill the NeilMed bottle to the fill line with distilled water at body temperature
-
Add the NeilMed saline packet and dissolve completely
-
Add the dissolved Manuka honey solution
-
Add the xylitol and mix gently until dissolved
-
Add the Johnson’s Baby Shampoo last — gently tip the bottle to mix without creating excessive foam
-
Cap and invert gently twice to combine
How to use:
-
Use twice daily — morning and evening — consistently
-
Standard NeilMed rinse technique — lean forward over the sink, head tilted, squeeze gently
-
Do not use tap water at any step — tap water in Scottsdale and the Phoenix area carries chlorine and minerals that will alter the formulation and should never be used for nasal irrigation due to infection risk
-
Prepare fresh for each use — do not store mixed solution
-
Continue for a minimum of four weeks before reassessing symptoms
Important: This protocol is appropriate for adults with documented recurrent sinusitis. If you have a known allergy to any component — particularly bee products for Manuka honey — do not use this formulation without physician guidance. Patients with nasal septal perforations should not perform nasal irrigation. If your symptoms include high fever, severe facial pain, visual changes, or neurological symptoms, seek immediate medical evaluation rather than beginning this protocol.
When This Protocol Is Not Enough
The holistic rinse protocol is a first-line biofilm disruption strategy — appropriate for patients in the RARS category who are caught in the urgent care antibiotic cycle and want to try a rational non-antibiotic approach before escalating care. It is not a substitute for culture-directed diagnosis and treatment in patients with established CRS, prior sinus surgery, or resistant organisms confirmed on culture.
If four weeks of consistent twice-daily holistic rinse does not produce meaningful improvement in your symptoms — measured by reduced congestion, reduced post-nasal drainage, fewer days of facial pressure — the next step is a MicroGenDX nasal microbiome evaluation.
MicroGenDX: The Test That Changes Everything
MicroGenDX is not a standard culture. It is next-generation DNA sequencing applied directly to nasal specimens — identifying every organism present in your sinus microbiome by its genetic fingerprint rather than its ability to grow on a culture plate. This distinction matters enormously. Standard cultures miss slow-growing organisms, anaerobes, and biofilm-embedded bacteria that are metabolically dormant during the culture period. MicroGenDX identifies them all — including the organisms most likely to be driving treatment-resistant disease.
What MicroGenDX tells you that no other test currently provides: the complete bacterial and fungal community in your sinuses, quantified by relative abundance; the specific antibiotic sensitivity patterns of your identified organisms; the presence of biofilm-associated species — Staphylococcus aureus, Pseudomonas aeruginosa, and others — whose resistance profiles change the entire treatment calculus; and the polymicrobial complexity that explains why a single-organism-targeted antibiotic course fails even when the primary organism is correctly identified.
The result guides culture-directed compound antibiotic rinse therapy — the specific antibiotic active against your identified organism, compounded by a pharmacy and delivered topically directly to the sinus mucosa. This bypasses first-pass hepatic metabolism entirely. The drug reaches the sinus mucosa at therapeutic concentration without passing through the gut — which means no disruption to your gut microbiome, and drug concentrations at the target site that oral dosing cannot achieve.
MicroGenDX is the laboratory I use at SAWC for this reason. The data behind next-generation sequencing in rhinology is compelling — standard cultures identify an organism in fewer than half of CRS specimens, while NGS sequencing identifies clinically significant organisms in the vast majority. If you want to understand more about how this test works and why it changes the treatment decision, I have written a dedicated article on MicroGenDX for the Airway & Sinus Wellness Review that covers it in full clinical detail.
And if culture-directed topical therapy does not achieve lasting resolution — if the biofilm has become too established, the sinus anatomy is compromised, or the ostia are chronically blocked — that is the conversation about surgical intervention: balloon sinuplasty to restore drainage, with intraoperative culture and direct inoculation of therapeutic agents into the newly opened sinus cavity at the moment of maximum accessibility.
Each step is rational. Each step is informed by what the previous step revealed. And for many patients — the ones who have been bouncing through urgent care and primary care getting the same prescription for years — the holistic rinse is the first time anyone has actually tried to address the biological reason their infection keeps coming back.
Dr. G’s Clinical Pearls
-
Biofilm-forming bacteria are present in over 75% of CRS patients by some published estimates — and in even higher proportions in patients who have had prior surgery or multiple antibiotic courses. If your patient has had three or more antibiotic courses, assume biofilm until proven otherwise.
-
Bacteria inside a biofilm are up to 1,000 times more resistant to antibiotics than planktonic bacteria. An oral antibiotic course reaching standard serum concentrations cannot penetrate a mature sinonasal biofilm at therapeutic levels.
-
The holistic rinse disrupts the biofilm mechanically and chemically before any antimicrobial effect is needed — xylitol disrupts matrix adhesion and quorum sensing, JBS dissolves the lipid membrane, and Manuka honey kills or washes away the exposed organisms.
-
Manuka honey rating matters. UMF 16+ or MGO 400+ from New Zealand is not interchangeable with regular honey or lower-rated products. The active compound — methylglyoxal — must be present at therapeutic concentration. Generic substitutions should not be made.
-
The organisms I am culturing from CRS patients presenting with acute exacerbations — Citrobacter, Klebsiella aerogenes, MRSA-risk Staph aureus, C. acnes — are not organisms that amoxicillin or azithromycin from urgent care was designed to treat. Culture-directed therapy is not optional in this population. It is necessary.
-
The 2025 AAO-HNS guideline recommends saline nasal irrigation as primary therapy for CRS. The holistic rinse protocol is an evidence-informed extension of this recommendation — adding biofilm-disrupting and antimicrobial components to the basic saline rinse that guidelines already endorse.
-
Every patient who comes to me with four or more sinus infections per year gets a conversation about what is living in their sinuses, why oral antibiotics cannot reach it, and what a rational escalation pathway looks like from holistic rinse through culture-directed compound rinse to surgical intervention if needed. That conversation is the one they have never had — and it is the one that finally makes sense of everything that has happened to them.
References
-
Payne SC, McKenna MK, et al. Clinical Practice Guideline: Adult Sinusitis Update. Otolaryngology–Head and Neck Surgery. 2025;173(Suppl). doi:10.1002/ohn.1344
-
Tong CCL, Woodworth BA, Palmer JN. Biofilms in Chronic Rhinosinusitis. In: Önerci Celebi Ö, Önerci TM, eds. Nasal Polyposis and its Management. Springer, Cham; 2024. doi:10.1007/978-3-031-33199-2_11
-
Prince AA, Steiger JD, Khalid AN, et al. Prevalence of biofilm-forming bacteria in chronic rhinosinusitis. Am J Rhinol. 2008;22(3):239-245. doi:10.2500/ajr.2008.22.3180
-
Tatar EC, Tatar I, Ocal B, et al. Prevalence of biofilms and their response to medical treatment in chronic rhinosinusitis without polyps. Otolaryngol Head Neck Surg. 2012;146(4):669-675. PMID:22241786
-
Psaltis AJ, Ha KR, Beule AG, Tan LW, Wormald PJ. Bacterial biofilms in patients with chronic rhinosinusitis — distribution and prevalence. Am J Rhinol Allergy. 2015;29(6):e190-e194. PMID:26406922
-
Paul NA, Ramesh Bhat M, Antony B, Jayaraman J. Antibiotic resistance and biofilm formation in Cutibacterium acnes: A descriptive cross-sectional study. Indian J Dermatol Venereol Leprol. 2025;91:315-320. doi:10.25259/IJDVL_539_2024
-
Beig M, Shirazi O, Ebrahimi E, et al. Prevalence of antibiotic-resistant Cutibacterium acnes isolates, a systematic review and meta-analysis. J Glob Antimicrob Resist. 2024;39:82-91. doi:10.1016/j.jgar.2024.07.005
-
Annavajhala MK, Gomez-Simmonds A, Uhlemann AC. Multidrug-Resistant Enterobacter cloacae Complex Emerging as a Global, Diversifying Threat. Front Microbiol. 2019;10:44. PMID:30778342
-
Tamma PD, Aitken SL, Bonomo RA, et al. IDSA Guidance on the Treatment of Antimicrobial-Resistant Gram-Negative Infections. Infectious Diseases Society of America; 2024. Available at: idsociety.org
-
Brook I. Microbiology of chronic rhinosinusitis. Eur J Clin Microbiol Infect Dis. 2016;35(7):1059-1068. doi:10.1007/s10096-016-2640-x
-
Mancuso G, Midiri A, Gerace E, Biondo C. Bacterial Antibiotic Resistance: The Most Critical Pathogens. Pathogens. 2021;10(10):1310. PMID:34684258
-
Cho SY, Choi SM, et al. Antimicrobial resistance patterns of Enterobacter cloacae and Klebsiella aerogenes strains isolated from clinical specimens: A twenty-year surveillance study. Microorganisms. 2023;11(4):902. doi:10.3390/microorganisms11040902
Want to Understand More?
This post is part of the Why Sinus Treatments Fail — And What Starts Before Them series on the Airway & Sinus Wellness Review. Every article below is free to read — and if you find them useful, subscribing brings new clinical education to your inbox every week.
On antibiotics, resistance, and why treatment keeps failing:
→ Why Antibiotics Keep Failing Your Sinus Infection
On MicroGenDX and culture-directed therapy:
→ What Is MicroGenDX — And Why Does It Change How We Treat Sinus Infections?
On balloon sinuplasty — the next step when rinse therapy isn’t enough:
→ Does Balloon Sinuplasty Actually Work?
→ Is Balloon Sinuplasty Painful? Here Is What the Procedure Actually Feels Like
→ Will Balloon Sinuplasty Correct My Post-Nasal Drainage?
On what your sinus infection is doing to the rest of your body:
→ Could My Husband’s Dementia Actually Be Brain Fog From Sinus Infections?
On understanding your diagnosis — sinusitis vs. allergies:
→ Is It Sinusitis or Allergies? How Do I Know Which One I Have?
On tooth pain, sinus pain, and the connection most patients miss:
→ Can a Tooth Infection Cause a Sinus Infection?
On living in Scottsdale and the Arizona desert environment:
→ Why Does Scottsdale’s Dry Heat Make My Sinuses So Much Worse?
On safe rinse products and NeilMed:
→ Is the NeilMed Sinus Rinse Safe? What You Need to Know Before You Use It
→ Browse the full Airway & Sinus Wellness Review — all articles, all FAQs, free to read
Why Sinus Treatments Fail — And What Starts Before Them — Patient education from the Sinus & Allergy Wellness Center of North Scottsdale.
About the Author
Franklyn R. Gergits, DO, MBA, FAOCO is an otolaryngologist and rhinologist with over 30 years of clinical experience. He is the founder of the Sinus & Allergy Wellness Center of North Scottsdale, where he performs in-office balloon sinuplasty, turbinate reduction, NEUROMARK®, and swell body reduction procedures under local anesthesia. He performed the first balloon sinuplasty in Pennsylvania, holds dual Entellus Centers of Excellence certifications, and is the originator of the Posterior Sinonasal Syndrome (PSS) hypothesis, with a preprint available at Preprints.org (DOI: 10.20944/preprints202603.0858.v1). ORCID: 0009-0000-4893-6332.
SinusAndAllergyWellnessCenter.com · 480-525-8999
This content is for educational purposes only and does not constitute medical advice. The organisms described represent clinical examples and should not be interpreted as findings applicable to all patients. If you are experiencing sinus symptoms, please consult a qualified physician for evaluation, culture-directed diagnosis, and individualized treatment recommendations.
Disclaimer:
The information provided in this article is for informational and educational purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease or medical condition. Always seek the guidance of your physician or other qualified healthcare provider with any questions you may have regarding a medical condition or treatment.
Results may vary: Treatment outcomes and health experiences may differ based on individual medical history, condition severity, and response to care.
Emergency Notice: If you are experiencing a medical emergency, call 911 or seek immediate medical attention.