Chapter 7: The Immune Era
Why Sinus Treatments Fail — And What Starts Before Them · Chapter 7 of the Series
Keywords: Chronic Rhinosinusitis (CRS), Immune Endotypes, Type 2 Inflammation, Posterior Sinonasal Inflammation, Eustachian Tube Dysfunction, Eustachian Tube Balloon Dilation, Unified Airway Disease, Biologic Therapy, Dupilumab, Mepolizumab, Omalizumab, Refractory CRS, Nasal Polyposis, Sublingual Immunotherapy (SLIT), Subcutaneous Immunotherapy (SCIT), Allergy Desensitization, Laryngopharyngeal Reflux (LPR), Pepsin, Posterior Symptom Burden, Neurogenic Inflammation, Posterior Nasal Nerve Treatment.
From Frustration to Clarity: Why Patients Kept Coming Back
By the early 2010s, rhinology had reached technical and diagnostic sophistication unmatched in its history. And yet, a familiar frustration persisted.
A subset of patients continued to return — not because anatomy had failed, but because they were still symptomatic. These patients did not complain primarily of classic obstruction. Instead, they described chronic posterior nasal drainage, throat clearing, globus sensation, chronic cough, ear fullness and pressure, intermittent hoarseness, facial pressure without radiographic obstruction, and fatigue disproportionate to imaging findings.
This disconnect forced an uncomfortable realization: anatomy had been optimized, imaging looked acceptable, but inflammation persisted — particularly posteriorly.
Posterior disease behaved differently than classic sinus obstruction. Symptoms fluctuated with environment, meals, sleep position, and season rather than imaging severity. This variability suggested physiologic regulation rather than mechanical blockage.
The field that had spent decades perfecting its tools was now confronting the limits of what those tools could explain. The patients who were not getting better were not outliers. They were data. And data, honestly interpreted, demands a new framework.
Immune Endotypes: Chronic Rhinosinusitis Is Not One Disease
The most intellectually significant development of this era was the recognition that Chronic Rhinosinusitis (CRS) is not a single condition. It is a heterogeneous group of disease processes unified by overlapping symptoms but driven by fundamentally different inflammatory mechanisms.
Endotype research — the classification of disease by underlying biological mechanism rather than surface presentation — revealed that CRS patients fall into distinct inflammatory categories. Type 2 inflammation, driven by Interleukin-4 (IL-4), Interleukin-5 (IL-5), and Interleukin-13 (IL-13), characterized the patient population with nasal polyps, tissue eosinophilia, and frequent comorbid asthma. Non-Type 2 inflammation — neutrophilic, mixed, or insufficiently characterized — drove a separate population whose disease behaved entirely differently.
This distinction was not academic. It had direct clinical consequences. A patient with Type 2 eosinophilic CRS with Nasal Polyps (CRSwNP) responded to steroids and eventually to biologics. A patient with neutrophilic non-Type 2 CRS did not — and the entire treatment framework that worked for the first patient was largely irrelevant for the second.
The European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS 2020) formalized this framework into clinical practice, providing the most comprehensive evidence-based guidance the field had yet produced. For the first time, treatment algorithms were organized not just by symptom duration but by biological disease type. The era of one-size-fits-all CRS management was formally over.
The field had been treating CRS as one disease for decades. It was not. It never had been.
Figure 1 illustrates the fundamental difference between Type 2 and non-Type 2 Chronic Rhinosinusitis — two distinct disease processes that share overlapping symptoms but require entirely different treatment approaches. Understanding which endotype drives a patient’s disease is the first step toward effective management.
The Unified Airway: Sinuses, Ears, and Lungs as One System
Running parallel to the endotype revolution was a conceptual shift that had profound implications for how the entire airway was understood and treated.
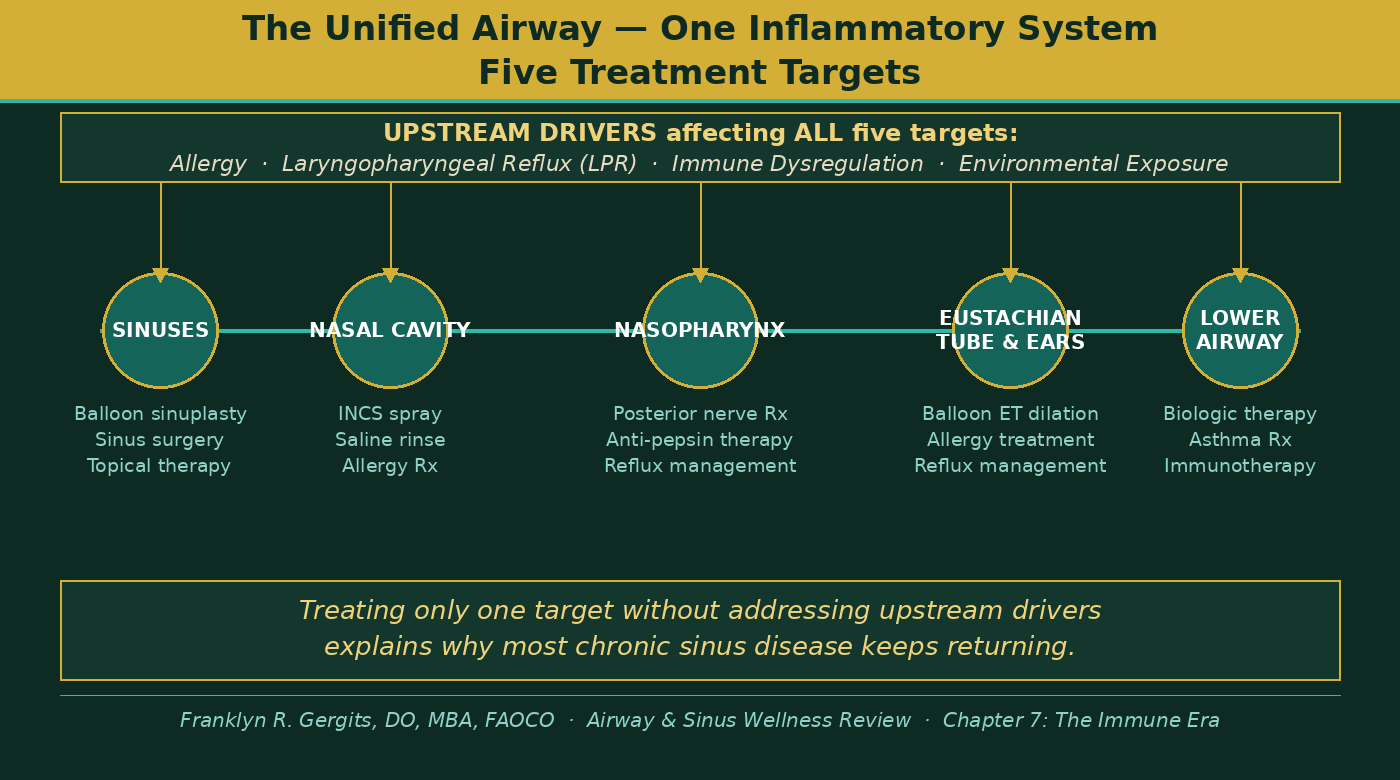
The unified airway model — the recognition that the nasal cavity, paranasal sinuses, Eustachian (auditory) tubes, middle ears, and lower airways share not just anatomical continuity but a common inflammatory biology — moved from theoretical framework to clinical standard during this era. The Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines formalized the epidemiological and pathophysiological links between allergic rhinitis and asthma. The clinical implication was direct: a physician treating sinusitis without addressing lower airway inflammation, and a physician treating asthma without addressing upper airway disease, were each treating half of a connected system and wondering why their results were incomplete.
This unified perspective changed how we thought about allergy treatment in sinus patients specifically. Allergic sensitization is not a separate diagnosis that coincidentally coexists with sinusitis — it is frequently the upstream inflammatory driver maintaining mucosal edema, obstructing sinus drainage, and perpetuating the cycle of recurrent infection. Treating anatomy without treating allergy is opening a door that inflammation will close again.
The ears complete the circuit. Eustachian tube dysfunction — the inability of the auditory tube to equalize middle ear pressure, ventilate the middle ear space, and drain accumulated fluid — is driven by the same upstream inflammatory forces that drive sinusitis. Allergic inflammation, posterior nasal mucosal edema, and reflux-driven nasopharyngeal irritation all impair Eustachian tube function. A patient whose ears feel chronically full and pressured is often a patient whose sinus and allergy disease has never been fully addressed. The ear is not a separate problem. It is a downstream expression of the same upstream biology.
Figure 2 shows the five connected treatment targets of the unified airway — from the sinuses to the lower airway — and the upstream drivers that affect all five simultaneously. Addressing one target in isolation while leaving upstream drivers untreated is the most common reason chronic airway disease keeps returning.
Allergy Immunotherapy: Treating the Root, Not the Branch
No discussion of this era is complete without acknowledging the maturation of allergy immunotherapy as a foundational component of long-term CRS management.
Subcutaneous Immunotherapy (SCIT) — the traditional allergy injection series — had been available for decades. But its role in CRS management had been underemphasized in surgical-era rhinology, where anatomy correction was the primary focus. The immune era changed that calculus. As endotype research clarified that allergic sensitization was a primary driver of mucosal inflammation in a substantial subset of CRS patients, immunotherapy moved from adjunct to essential.
Sublingual Immunotherapy (SLIT) — allergen drops administered under the tongue — expanded access to desensitization for patients who could not commit to the office-based injection schedule required by SCIT. The World Allergy Organization (WAO) position paper formalized SLIT’s evidence base, and multiple meta-analyses confirmed its efficacy for allergic rhinitis and its impact on lower airway disease. For many patients, particularly those in high-allergen environments like Arizona’s extended pollen seasons, immunotherapy became the single most important long-term intervention available — not because it relieved symptoms acutely, but because it gradually reduced the immune system’s reactivity to the triggers driving chronic mucosal inflammation.
The clinical reality at SAWC reflects this. Patients who complete a full course of allergy immunotherapy consistently demonstrate reduced frequency of sinus exacerbations, reduced dependence on rescue medications, and improved long-term mucosal health on endoscopy. The anatomy may have been corrected years earlier. The immunotherapy is what keeps the mucosal environment stable enough for that correction to hold.
Treating allergy is not treating the symptom. It is treating the driver.
Neurogenic Inflammation: When the Airway Was Following Instructions
As posterior symptoms were studied more carefully, a pattern emerged that challenged the purely structural model of sinus disease.
Not all mucus was inflammatory exudate. Not all drainage was infectious. Not all swelling was structural.
Many patients were producing secretions because the airway was being instructed to produce them.
The posterior nasal cavity contains dense parasympathetic innervation responsible for humidification, glandular secretion, and mucosal protection. Under chronic stimulation that innervation becomes pathologic. Irritants — inflammatory cytokines, allergens, refluxed gastric contents, temperature change, and environmental triggers — activate sensory receptors within the posterior nasal mucosa. These signals propagate through parasympathetic pathways and trigger glandular hypersecretion that has nothing to do with infection.
The airway was not blocked. It was over-responding.
This explained why some patients with minimal objective disease remained profoundly symptomatic. Their inflammation was being perpetuated by reflex circuitry rather than infection or anatomy. Treating the infection did nothing. Treating the anatomy did nothing. The signal had to be addressed at its source.
Office-based technology changed the landscape. Targeted treatment of the posterior nasal nerve — through cryotherapy and radiofrequency ablation — allowed localized modulation of parasympathetic input while preserving surrounding structures. Many patients who had undergone technically successful sinus procedures but remained symptomatic improved dramatically once this reflex pathway was addressed.
The disease in these individuals had never been obstruction. It had been instruction.
Eustachian Tube Balloon Dilation: Learning From a Pioneer
No discussion of this era is complete without addressing the ear.
For the sinus and airway clinician, the Eustachian tube had long been the missing link in the unified airway model. Patients with CRS frequently presented with coinciding ear symptoms — fullness, pressure, muffled hearing, and recurrent otitis media (middle ear infections) — that the sinus-focused clinical framework had no satisfying answer for. The management default was bilateral myringotomy with tympanostomy tube insertion — placing ear tubes in the tympanic membrane (eardrum) to restore middle ear ventilation. It worked. But it was a mechanical solution to what was increasingly understood to be a biological problem.
The tympanic membrane is not built for repeated tube insertion. Every procedure carries risk and every round of tubes compounds the cumulative burden on the eardrum over time.
Balloon dilation of the Eustachian tube offered a fundamentally different approach — treating the obstruction directly rather than bypassing it.
I had the privilege of attending the Endoscopic Surgery of the Sinuses, Eustachian Tube, and Ear Course at Harvard Medical School in 2015, 2016, and 2017 — three consecutive years during the period when Dr. Dennis Poe was conducting the FDA (Food and Drug Administration) clinical trials for Eustachian tube balloon dilation. My daughter was attending Boston University at the time, and I made a point of scheduling around this course each year we visited her in Boston. Those three years gave me something no published paper could: direct access to one of the most brilliant and generous physicians I have encountered in my career.
Dr. Poe is a remarkable clinician and educator. He navigated the FDA disclosure restrictions with complete professionalism, sharing everything that was open for educational purposes — the anatomy, the physiologic rationale, the patient selection principles, and the clinical promise of the technology. What resonated most strongly from his teaching was straightforward and clinically profound: continue with allergy, sinus, and reflux treatment. The Eustachian tube does not exist in isolation. It sits at the intersection of every upstream inflammatory driver — allergic disease, posterior sinonasal inflammation, and reflux — and treating those drivers is inseparable from achieving durable Eustachian tube outcomes.
Eustachian tube balloon dilation received FDA approval on September 19, 2016. Having attended those courses in the years surrounding that approval, I was positioned to bring this technology to community practice with a deeper understanding of its appropriate application than a physician who encountered it only after approval.
As an early adopter of balloon sinuplasty, I had already seen the impact of catheter-based dilation technology in the sinus — both in the operating room and in the office setting. The transition to Eustachian tube dilation was a natural clinical evolution. The same philosophy applied: restore the drainage pathway, reduce the mechanical obstruction, and allow the biology of the airway to normalize.
The clinical results have matched what Dr. Poe described. The patients obtaining balloon Eustachian tube dilation at SAWC are typically patients who have sinus troubles that coincide with ear and Eustachian tube dysfunction. Many patients do not realize their sinus disease is a contributing factor because their dominant complaint is ear symptoms — pressure, fullness, hearing changes — that they have never connected to what is happening in their sinuses. Addressing the complete picture — sinus, nose, Eustachian tube, and upstream inflammatory drivers — produces outcomes that treating the ear in isolation never could.
Today I place bilateral tympanostomy tubes very, very rarely. Balloon Eustachian tube dilation has replaced the vast majority of what I would previously have managed with repeated ear tube procedures. The tympanic membrane is better for it.
Biologics: When Immune Targeting Changed Everything
Perhaps the most definitive validation of CRS as an immune disease came not from a surgical advance or a diagnostic tool — but from a medication that had no business working as well as it did.
Dupilumab — a monoclonal antibody targeting the shared receptor for Interleukin-4 and Interleukin-13, the central cytokines of Type 2 inflammation — was originally developed for atopic dermatitis (eczema). When it was evaluated in patients with severe CRS with Nasal Polyps (CRSwNP), the results were striking. Polyp burden decreased. Smell returned. Nasal obstruction improved. Patients who had failed multiple surgeries and repeated courses of systemic corticosteroids (steroids) responded to a targeted immune intervention that had never set foot in the sinus.
The implication was unavoidable: in these patients, the sinus was not the source of the disease. It was the target. The disease was systemic. The inflammation was immune-mediated. Surgery had been opening anatomy that was being re-obstructed by biology that surgery could not address.
Dupilumab was followed by other biologics targeting the Type 2 inflammatory axis — mepolizumab targeting Interleukin-5 (IL-5), omalizumab targeting Immunoglobulin E (IgE) — each confirming in its own way that a subset of CRS was fundamentally a systemic immune disease expressing itself in the nose and sinuses. The Phase 3 LIBERTY NP SINUS trials established dupilumab’s efficacy and safety profile at a level sufficient for regulatory approval across multiple indications — atopic dermatitis, asthma, and CRSwNP — making it the first biologic to span the entire unified airway simultaneously.
For the first time, the historic non-responder subgroup — the patients who had failed anatomy correction, allergy management, and repeated antibiotic courses — had a therapy that addressed their disease at its biological source.
Not drainage. Not steroids alone. Not repeated surgery. Biology.
But even biologics did not explain everything. A meaningful proportion of CRS patients — including many with posterior-dominant symptoms, posterior nasal inflammation, and upstream reflux drivers — did not fit cleanly into the Type 2 endotype model. They did not have polyps. They did not have eosinophilia. They did not respond to biologic therapy. They had something else.
That something else is the subject of the final chapter.
What This Era Could Not Yet See
After thirty years in clinical practice — through the endoscopic revolution, the guideline era, the precision era, and now the immune era — I had watched rhinology develop extraordinary tools for the patients it could explain. The endotype model identified Type 2 CRS and gave it a treatment. The biologic era gave that treatment unprecedented efficacy. Eustachian tube dilation extended the reach of catheter-based technology to the ear. Immunotherapy addressed the allergic driver at its biological source.
And yet the patients who confounded me most were not the ones with polyps, or the ones with documented Type 2 eosinophilia, or the ones with clear anatomical obstruction. They were the ones with none of those findings — whose CT scans were normal or near-normal, whose endoscopy showed posterior mucosal changes that did not fit established disease categories, whose symptoms fluctuated with meals and sleep position and season in ways that suggested something upstream of the sinuses entirely.
These patients were not failing the field’s tools. The field’s tools were not designed to find what was driving them.
Something was priming the posterior sinonasal mucosa before the sinuses ever became involved. Something was generating inflammation that no antibiotic, no surgery, no steroid, and no biologic was designed to address. And that something — in my clinical observation across thousands of patient encounters — had a name, a mechanism, and a trajectory that the field had not yet formally described.
The final chapter of this series is the attempt to name it.
Dr. G’s Clinical Pearls — The Immune Era
-
Chronic Rhinosinusitis (CRS) is fundamentally an inflammatory immune disease in a significant subset of patients — not a structural or infectious problem
-
Anatomy restoration enables treatment but does not normalize disease biology
-
Endotype matters — Type 2 and non-Type 2 CRS require entirely different treatment frameworks
-
Steroid responsiveness often predicts the immune-driven CRS phenotype
-
Recurrent disease after ideal surgery signals biologic drivers rather than technical failure
-
The unified airway connects sinuses, Eustachian tubes, ears, and lungs — treating one in isolation misses the system
-
Allergy immunotherapy is not adjunctive care — it is upstream driver treatment and belongs at the center of long-term CRS management
-
The Eustachian tube sits at the intersection of every upstream airway inflammatory driver — allergy, posterior sinonasal inflammation, and reflux must all be addressed
-
Biologic therapy finally addresses the historic CRS non-responder subgroup — but only for the Type 2 endotype
-
The patients who still do not respond to biologics are telling us something the field has not yet fully heard
Reference List — Chapter 7
1. Fokkens WJ et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020 (EPOS 2020). Rhinology Suppl. 2020;29:1–464.
2. Tomassen P et al. Inflammatory endotypes of chronic rhinosinusitis based on cluster analysis of biomarkers. J Allergy Clin Immunol. 2016;137:1449–1456.
3. Bachert C, Han JK, Desrosiers M, et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps. Lancet. 2019;394:1638–1650.
4. Bachert C et al. LIBERTY NP SINUS-24 and SINUS-52 investigators. Dupilumab efficacy and safety in a combined analysis of Phase 3 trials. Allergy. 2020;75:1153–1165.
5. Gevaert P et al. Omalizumab is effective in allergic and nonallergic patients with nasal polyps and asthma. J Allergy Clin Immunol. 2013;131:110–116.
6. Han JK et al. Mepolizumab for chronic rhinosinusitis with nasal polyps. J Allergy Clin Immunol. 2019;143:193–201.
7. Bousquet J et al. Allergic Rhinitis and its Impact on Asthma (ARIA) 2008 update. Allergy. 2008;63(Suppl 86):8–160.
8. Hens G, Hellings PW. The nose: gatekeeper and trigger of bronchial disease. Rhinology. 2006;44:179–187.
9. Canonica GW et al. Sub-lingual immunotherapy: World Allergy Organization position paper 2013 update. World Allergy Organ J. 2014;7:6.
10. Di Bona D et al. Efficacy of sublingual immunotherapy with grass allergens for seasonal allergic rhinitis: a systematic review and meta-analysis. J Allergy Clin Immunol. 2010;126:558–566.
11. Poe DS, Hanna BM. Balloon dilation of the cartilaginous Eustachian tube: a preliminary study in human cadavers. Otolaryngol Head Neck Surg. 2011;145:999–1003.
12. Poe DS et al. Balloon dilation of the Eustachian tube for dilatory dysfunction: a randomized controlled trial. Laryngoscope. 2018;128:1200–1206.
13. Huisman JML et al. Balloon dilation of the Eustachian tube: a systematic review. Otolaryngol Head Neck Surg. 2018;158:222–230.
14. FDA 510(k) clearance: Eustachian tube balloon dilation system. September 19, 2016.
15. Van Zele T et al. Differentiation of chronic sinus diseases by measurement of inflammatory mediators. Allergy. 2006;61:1280–1289.
16. Rudmik L, Soler ZM. Medical therapies for adult chronic rhinosinusitis: a systematic review. JAMA. 2015;314:926–939.
17. Rosenfeld RM et al. Clinical Practice Guideline (Update): Adult Sinusitis. Otolaryngol Head Neck Surg. 2015;152(2 Suppl):S1–S39.
18. Johnston N et al. Pepsin as a causal agent of inflammation during nonacidic reflux. Otolaryngol Head Neck Surg. 2004;130:649–652.
19. Crapko M et al. Role of extra-esophageal reflux in chronic otitis media with effusion. Laryngoscope. 2007;117:1419–1423.
20. Hellings PW et al. Uncontrolled allergic rhinitis and chronic rhinosinusitis: where do we stand today? Allergy. 2013;68:1–7.
21. Gergits FR. The Sinus Is the Victim, Pepsin Is the Perpetrator: Posterior Sinonasal Syndrome as the Etiological Precursor to Chronic Rhinosinusitis, Field Carcinogen of the Upper Aerodigestive Tract, and the Biological Imperative for Mucosal-Preserving Surgery. Preprints 2026. DOI: 10.20944/preprints202603.0858.v1.
Franklyn R. Gergits, DO, MBA, FAOCO
Otolaryngologist & Rhinologist | 30+ Years Clinical Experience
Founder, Sinus & Allergy Wellness Center of North Scottsdale
SinusAndAllergyWellnessCenter.com · 480-525-8999
ORCID: 0009-0000-4893-6332
Preprint: https://doi.org/10.20944/preprints202603.0858.v1
This content is for educational purposes only and does not constitute medical advice.
Disclaimer:
The information provided in this article is for informational and educational purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease or medical condition. Always seek the guidance of your physician or other qualified healthcare provider with any questions you may have regarding a medical condition or treatment.
Results may vary: Treatment outcomes and health experiences may differ based on individual medical history, condition severity, and response to care.
Emergency Notice: If you are experiencing a medical emergency, call 911 or seek immediate medical attention.